Back حلقي الهكسانون Arabic Sikloheksanon AZ سیکلوهقزانون AZB Цыклагексанон BE Ciclohexanona Catalan Cyklohexanon Czech Cyclohexanon German Cikloheksanono EO Ciclohexanona Spanish Ziklohexanona EU
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cyclohexanone | |||
| Other names
oxocyclohexane, pimelic ketone, ketohexamethylene, cyclohexyl ketone, ketocyclohexane, hexanon, Hydrol-O, Sextone, K, Anone
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.003.302 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H10O | |||
| Molar mass | 98.15 g/mol | ||
| Appearance | Colorless liquid[3] | ||
| Odor | Peppermint or acetone-like | ||
| Density | 0.9478 g/mL, liquid | ||
| Melting point | −47 °C (−53 °F; 226 K)[6] | ||
| Boiling point | 155.65 °C (312.17 °F; 428.80 K) | ||
| 8.6 g/100 mL (20 °C) | |||
| Solubility in all organic solvents | Miscible | ||
| log P | 0.81 | ||
| Vapor pressure | 5 mmHg (20 °C)[4] | ||
| −62.04·10−6 cm3/mol | |||
Refractive index (nD)
|
1.447 | ||
| Viscosity | 2.02 cP at 25 °C[5] | ||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
+229.03 J·K−1·mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
−270.7 kJ·mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−3519.3 kJ·mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
  
| |||
| Danger[7] | |||
| H226, H302, H305, H312, H315, H318, H332[7] | |||
| P280, P305+P351+P338[7] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 44 °C (111 °F; 317 K) | ||
| 420 °C (788 °F; 693 K) | |||
| Explosive limits | 1.1–9.4% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
1200 mg/kg (cat, orally); 2362 mg/kg (rat, orally)[8] | ||
LC50 (median concentration)
|
8000 ppm (rat, 4 hr)[9] | ||
LCLo (lowest published)
|
4706 ppm (mouse, 1.5 hr)[9] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 50 ppm (200 mg/m3)[4] | ||
REL (Recommended)
|
TWA 25 ppm (100 mg/m3) [skin][4] | ||
IDLH (Immediate danger)
|
700 ppm[4] | ||
| Related compounds | |||
Related ketones
|
Cyclopentanone, cycloheptanone | ||
Related compounds
|
Cyclohexanol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
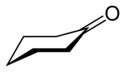
Cyclohexanone is the organic compound with the formula (CH2)5CO. The molecule consists of six-carbon cyclic molecule with a ketone functional group. This colorless oily liquid[3] has a sweet odor reminiscent of benzaldehyde. Over time, samples of cyclohexanone assume a pale yellow color.[10]
Cyclohexanone is slightly soluble in water and miscible with common organic solvents. Millions of tonnes are produced annually, mainly as a precursor to nylon.[11]
- ^ "ICSC 0425 – CYCLOHEXANONE". inchem.org. Retrieved 2022-08-24.
- ^ "CDC – NIOSH Pocket Guide to Chemical Hazards – Cyclohexanone". Centers for Disease Control and Prevention (CDC). Retrieved August 24, 2022.
- ^ a b "Cyclohexanone (CID 7967)". PubChem.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0166". National Institute for Occupational Safety and Health (NIOSH).
- ^ Ch. Wohlfarth. "Viscosity of cyclohexanone". In M.D. Lechner (ed.). Viscosity of Pure Organic Liquids and Binary Liquid Mixtures · Supplement to IV/18. Springer-Verlag. doi:10.1007/978-3-540-75486-2_192.
- ^ "Cyclohexanone". Sigma-Aldrich.
- ^ a b c Sigma-Aldrich Co., Cyclohexanone.
- ^ Cyclohexanone: toxicity for animals and humans
- ^ a b "Cyclohexanone". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Cyclohexanone (CID 7967)". PubChem.
- ^ Musser, Michael T. (October 15, 2011). "Cyclohexanol and Cyclohexanone". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KgaA. doi:10.1002/14356007.a08_217.pub2. ISBN 978-3-527-30673-2. OCLC 46878292. Retrieved January 27, 2009.