Our website is made possible by displaying online advertisements to our visitors.
Please consider supporting us by disabling your ad blocker.
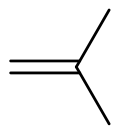
Isobutylene
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methylprop-1-ene | |||
| Other names
2-Methylpropene
Isobutene γ-Butylene 2-Methylpropylene Methylpropene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.003.697 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1055 In Liquefied petroleum gas: 1075 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties[1] | |||
| C4H8 | |||
| Molar mass | 56.106 g/mol | ||
| Appearance | Colorless gas | ||
| Density | 0.5879 g/cm3, liquid | ||
| Melting point | −140.3 °C (−220.5 °F; 132.8 K) | ||
| Boiling point | −6.9 °C (19.6 °F; 266.2 K) | ||
| -44.4·10−6 cm3/mol | |||
| Hazards[2] | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H220 | |||
| P210, P377, P381, P403 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | flammable gas | ||
| 465 °C (869 °F; 738 K) | |||
| Explosive limits | 1.8–9.6% | ||
| Related compounds | |||
Related butenes
|
1-Butene cis-2-Butene trans-2-Butene | ||
Related compounds
|
Isobutane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Isobutylene (or 2-methylpropene) is a hydrocarbon with the chemical formula (CH3)2C=CH2. It is a four-carbon branched alkene (olefin), one of the four isomers of butylene. It is a colorless flammable gas, and is of considerable industrial value.[3]
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (11th ed.). Merck. 1989. ISBN 091191028X., 5024.
- ^ Isobutene, International Chemical Safety Card 1027, Geneva: International Programme on Chemical Safety, April 2000
- ^ Geilen, Frank M.A.; Stochniol, Guido; Peitz, Stephan; Schulte-Koerne, Ekkehard (2014). "Butenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a04_483.pub3. ISBN 978-3-527-30673-2.
Previous Page Next Page





