Our website is made possible by displaying online advertisements to our visitors.
Please consider supporting us by disabling your ad blocker.
Glatiramer acetate
 | |
| Clinical data | |
|---|---|
| Trade names | Copaxone,[1] Glatopa,[2] Brabio |
| Other names | Copolymer 1, Cop-1 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603016 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous injection |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.248.824 |
| Chemical and physical data | |
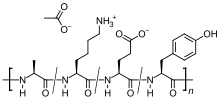
| Formula | C25H45N5O13 |
| Molar mass | 623.657 g·mol−1 |
| | |
Glatiramer acetate, sold under the brand name Copaxone among others, is an immunomodulator medication used to treat multiple sclerosis.[1][2] Glatiramer acetate is approved in the United States to reduce the frequency of relapses, but not for reducing the progression of disability. Observational studies, but not randomized controlled trials, suggest that it may reduce progression of disability. While a conclusive diagnosis of multiple sclerosis requires a history of two or more episodes of symptoms and signs, glatiramer acetate is approved to treat a first episode anticipating a diagnosis. It is also used to treat relapsing-remitting multiple sclerosis. It is administered by subcutaneous injection.[1][2]
It is a mixture of random-sized peptides that are composed of the four amino acids found in myelin basic protein, namely glutamic acid, lysine, alanine, and tyrosine. Myelin basic protein is the antigen in the myelin sheaths of the neurons that stimulates an autoimmune reaction in people with MS, so the peptide may work as a decoy for the attacking immune cells.
Glatiramer acetate was originally discovered at the Weizmann Institute of Science. It is on the World Health Organization's List of Essential Medicines.[6]
- ^ a b c d "Copaxone- glatiramer acetate injection, solution". DailyMed. 23 July 2020. Archived from the original on 31 October 2021. Retrieved 11 November 2020.
- ^ a b c d "Glatopa- glatiramer acetate injection, solution". DailyMed. 31 July 2020. Archived from the original on 17 November 2020. Retrieved 11 November 2020.
- ^ "Neurological therapies". Health Canada. 9 May 2018. Archived from the original on 4 December 2024. Retrieved 13 April 2024.
- ^ "Brabio 20 mg/mL Solution for Injection, Pre-filled Syringe - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 17 November 2020. Retrieved 11 November 2020.
- ^ "Copaxone 20 mg/ml solution for injection in pre-filled syringe - Summary of Product Characteristics (SmPC)". (emc). 29 September 2020. Archived from the original on 12 November 2020. Retrieved 11 November 2020.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
Previous Page Next Page


