Our website is made possible by displaying online advertisements to our visitors.
Please consider supporting us by disabling your ad blocker.
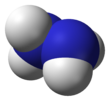
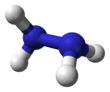
Hydrazine
| |||
| |||
 Anhydrous hydrazine
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydrazine[2]
| |||
| Systematic IUPAC name
Diazane[2] | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 878137 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.560 | ||
| EC Number |
| ||
| 190 | |||
| KEGG | |||
| MeSH | Hydrazine | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2029 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| N2H4 | |||
| Molar mass | 32.0452 g/mol | ||
| Appearance | Colorless, fuming, oily liquid[3] | ||
| Odor | Ammonia-like[3] | ||
| Density | 1.021 g/cm3 | ||
| Melting point | 2 °C; 35 °F; 275 K | ||
| Boiling point | 114 °C; 237 °F; 387 K | ||
| Miscible[3] | |||
| log P | 0.67 | ||
| Vapor pressure | 1 kPa (at 30.7 °C) | ||
| Acidity (pKa) | 8.10 ([N2H5]+)[4] | ||
| Basicity (pKb) | 5.90 | ||
| Conjugate acid | Hydrazinium | ||
Refractive index (nD)
|
1.46044 (at 22 °C) | ||
| Viscosity | 0.876 cP | ||
| Structure | |||
| Triangular pyramidal at N | |||
| 1.85 D[5] | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
121.52 J/(K·mol) | ||
Std enthalpy of
formation (ΔfH⦵298) |
50.63 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
    
| |||
| Danger | |||
| H226, H301, H311, H314, H317, H331, H350, H410 | |||
| P201, P261, P273, P280, P301+P310, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 52 °C (126 °F; 325 K) | ||
| 24 to 270 °C (75 to 518 °F; 297 to 543 K) | |||
| Explosive limits | 1.8–100% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
59–60 mg/kg (oral in rats, mice)[6] | ||
LC50 (median concentration)
|
260 ppm (rat, 4 h) 630 ppm (rat, 1 h) 570 ppm (rat, 4 h) 252 ppm (mouse, 4 h)[7] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 1 ppm (1.3 mg/m3) [skin][3] | ||
REL (Recommended)
|
Ca C 0.03 ppm (0.04 mg/m3) [2-hour][3] | ||
IDLH (Immediate danger)
|
Ca [50 ppm][3] | ||
| Safety data sheet (SDS) | ICSC 0281 | ||
| Related compounds | |||
Other anions
|
Tetrafluorohydrazine Hydrogen peroxide Diphosphane Diphosphorus tetraiodide | ||
Other cations
|
Organic hydrazines | ||
Related Binary azanes
|
Ammonia Triazane | ||
Related compounds
|
Diazene Triazene Tetrazene Diphosphene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrazine is an inorganic compound with the chemical formula N2H4. It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly hazardous unless handled in solution as, for example, hydrazine hydrate (N2H4·xH2O).
Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to pharmaceuticals and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion.[8] As of 2000[update], approximately 120,000 tons of hydrazine hydrate (corresponding to a 64% solution of hydrazine in water by weight) were manufactured worldwide per year.[9]
Hydrazines are a class of organic substances derived by replacing one or more hydrogen atoms in hydrazine by an organic group.[9]
- ^ "NIOSH Guide—Hydrazine". Centers for Disease Control. Retrieved 16 Aug 2012.
- ^ a b "hydrazine—PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0329". National Institute for Occupational Safety and Health (NIOSH).
- ^ Hall HK, et al. (1957). "Correlation of the Base Strengths of Amines1". J. Am. Chem. Soc. 79 (20): 5441. doi:10.1021/ja01577a030.
- ^ Greenwood NN, Earnshaw A (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Martel B, Cassidy K, et al. (2004). Chemical Risk Analysis: A Practical Handbook. Amsterdam: Butterworth–Heinemann. p. 361. ISBN 978-1-903996-65-2. OCLC 939257974.
- ^ "Hydrazine". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Tsubakizaki S, Takada M, Gotou H, Mawatari K, Ishihara N, Kai R (2009). "Alternatives to Hydrazine in Water Treatment at Thermal Power Plants" (PDF). Mitsubishi Heavy Industries Technical Review. 6 (2): 43–47.
- ^ a b Cite error: The named reference
Ullmannwas invoked but never defined (see the help page).
Previous Page Next Page