Our website is made possible by displaying online advertisements to our visitors.
Please consider supporting us by disabling your ad blocker.
Phthalic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzene-1,2-dicarboxylic acid | |
| Other names
1,2-Benzenedioic acid
Phthalic acid Benzene-1,2-dioic acid ortho-Phthalic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.703 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H6O4 | |
| Molar mass | 166.132 g/mol |
| Appearance | white solid |
| Density | 1.593 g/cm3, solid |
| Melting point | 207 °C (405 °F; 480 K)[3] |
| 0.6 g / 100 mL [1] | |
| Acidity (pKa) | 2.89, 5.51[2] |
| -83.61·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related carboxylic acids
|
Isophthalic acid Terephthalic acid |
Related compounds
|
Phthalic anhydride Phthalimide Phthalhydrazide Phthaloyl chloride Benzene-1,2- dicarboxaldehyde |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
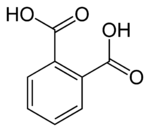
In organic chemistry, phthalic acid is an aromatic dicarboxylic acid, with formula C6H4(CO2H)2 and structure HO(O)C−C6H4−C(O)OH. Although phthalic acid is of modest commercial importance, the closely related derivative phthalic anhydride is a commodity chemical produced on a large scale.[4] Phthalic acid is one of three isomers of benzenedicarboxylic acid, the others being isophthalic acid and terephthalic acid.
- ^ "PHTHALIC ACID". hazard.com.
- ^ Brown, H.C., et al., in Baude, E.A. and Nachod, F.C., Determination of Organic Structures by Physical Methods, Academic Press, New York, 1955.
- ^ Several melting points are reported, for example: (i) 480. K (NIST website), (ii) 210−211 °C with decomposition (Sigma-Aldrich on-line), (iii) 191 °C in a sealed tube (Ullmann's Encyclopedia of Industrial Chemistry), (iv) 230 °C with conversion to phthalic anhydride and water (J.T.Baker MSDS).
- ^ Lorz, Peter M.; Towae, Friedrich K.; Enke, Walter; et al. (2007). "Phthalic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a20_181.pub2. ISBN 978-3-527-30673-2.
Previous Page Next Page



