
Back محب للنواة Arabic Nukleofil BS Nucleòfil Catalan Nukleofil Czech Niwcleoffil CY Nukleofil Danish Nukleophilie German Πυρηνόφιλο Greek Nucleófilo Spanish Nukleofiil ET
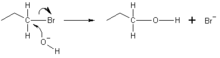
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases.
Nucleophilic describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. The difference between the two is, that basicity is a thermodynamic property (i.e. relates to an equilibrium state), but nucleophilicity is a kinetic property, which relates to rates of certain chemical reactions.[1]
- ^ Uggerud, Einar (2006-01-23). "Nucleophilicity—Periodic Trends and Connection to Basicity". Chemistry – A European Journal. 12 (4): 1127–1136. doi:10.1002/chem.200500639. ISSN 0947-6539.