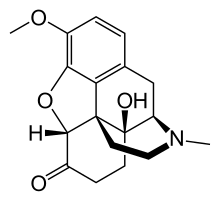
 | |
 | |
| Combination of | |
|---|---|
| Oxycodone | Opioid analgesic |
| Naloxone | Opioid receptor antagonist |
| Clinical data | |
| Trade names | Targin, Targiniq, Targinact, others |
| AHFS/Drugs.com | International Drug Names UK Drug Information |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| (verify) | |
Oxycodone/naloxone, sold under the brand name Targin among others, is a combination pain medication available as modified-release tablets administered by mouth.[5]
The oxycodone component is an opioid and is responsible for the pain-relieving effects. Naloxone, an opioid antagonist, opposes the effects of opioids but is poorly absorbed into the blood stream when administered orally; therefore, most of the dose remains in the gastrointestinal tract. This local presence reduces opioid-induced constipation by preventing oxycodone from binding to gut opioid receptors, without diminishing overall analgesic efficacy compared to oxycodone alone. A 2008 study demonstrated a significant reduction in constipation.[6] Oxycodone/naloxone was released in 2014 in the United States,[5] in 2006 in Germany, and has been available in some other European countries since 2009. In the United Kingdom, the 10 mg oxycodone / 5 mg naloxone and 20 mg / 10 mg strengths were approved in December 2008, and the 40 mg / 20 mg and 5 mg / 10 mg strengths received approval in July 2019.[7]
Preliminary evidence suggests that oxycodone/naloxone may be an effective treatment for severe, refractory restless legs syndrome if first-line therapies have not been effective.[8][9][10]
- ^ "Public Summary". www.ebs.tga.gov.au. Australian Government, Department of Health and Aged Care. Retrieved 20 May 2024.
- ^ "Oxonal (AU Pharma Pty Ltd)". Therapeutic Goods Administration (TGA). 28 September 2022. Archived from the original on 18 March 2023. Retrieved 9 April 2023.
- ^ "Targin Product information". Health Canada. 22 October 2009. Archived from the original on 29 April 2023. Retrieved 9 April 2023.
- ^ "Targinact 5 mg/2.5 mg prolonged-release tablets - Summary of Product Characteristics (SmPC)". (emc). 16 March 2021. Archived from the original on 10 April 2023. Retrieved 9 April 2023.
- ^ a b c "Targiniq ER- oxycodone hydrochloride/naloxone hydrochloride tablet, film coated, extended release". DailyMed. 22 December 2016. Archived from the original on 18 March 2023. Retrieved 18 March 2023.
- ^ Simpson K, Leyendecker P, Hopp M, Müller-Lissner S, Löwenstein O, De Andrés J, et al. (December 2008). "Fixed-ratio combination oxycodone/naloxone compared with oxycodone alone for the relief of opioid-induced constipation in moderate-to-severe noncancer pain". Current Medical Research and Opinion. 24 (12): 3503–3512. doi:10.1185/03007990802584454. PMID 19032132. S2CID 73061000. Archived from the original on 26 January 2013. Retrieved 9 April 2009.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Mundipharma (26 January 2009). "Targin (oral oxycodone/naloxone prolonged-release tablet) now launching across Europe to control severe chronic pain with significantly reduced risk of opioid-induced constipation". Archived from the original on 17 March 2009. Retrieved 9 April 2009.
- ^ de Biase S, Valente M, Gigli GL (2016). "Intractable restless legs syndrome: role of prolonged-release oxycodone-naloxone". Neuropsychiatric Disease and Treatment. 12: 417–425. doi:10.2147/NDT.S81186. PMC 4770072. PMID 26966363.
- ^ Trenkwalder C, Beneš H, Grote L, García-Borreguero D, Högl B, Hopp M, et al. (December 2013). "Prolonged release oxycodone-naloxone for treatment of severe restless legs syndrome after failure of previous treatment: a double-blind, randomised, placebo-controlled trial with an open-label extension". The Lancet. Neurology. 12 (12): 1141–1150. doi:10.1016/S1474-4422(13)70239-4. PMID 24140442. S2CID 35122538.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ de Oliveira CO, Carvalho LB, Carlos K, Conti C, de Oliveira MM, Prado LB, et al. (June 2016). "Opioids for restless legs syndrome". The Cochrane Database of Systematic Reviews. 2016 (6): CD006941. doi:10.1002/14651858.CD006941.pub2. PMC 6885031. PMID 27355187.
