Our website is made possible by displaying online advertisements to our visitors.
Please consider supporting us by disabling your ad blocker.
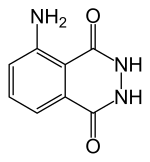
Luminol
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
5-Amino-2,3-dihydrophthalazine-1,4-dione | |
| Other names
5-Amino-2,3-dihydro-1,4-phthalazinedione
o-Aminophthaloyl hydrazide o-Aminophthalyl hydrazide 3-Aminophthalhydrazide 3-Aminophthalic hydrazide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.556 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H7N3O2 | |
| Molar mass | 177.16 g/mol |
| Melting point | 319 °C (606 °F; 592 K) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | MSDS for luminol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Luminol (C8H7N3O2) is a chemical that exhibits chemiluminescence, with a blue glow, when mixed with an appropriate oxidizing agent. Luminol is a white-to-pale-yellow crystalline solid that is soluble in most polar organic solvents but insoluble in water.
Forensic investigators use luminol to detect trace amounts of blood at crime scenes, as it reacts with the iron in hemoglobin. Biologists use it in cellular assays to detect copper, iron, and cyanides as well as specific proteins via western blotting.[2]
When luminol is sprayed evenly across an area, trace amounts of an activating oxidant make the luminol emit a blue glow that can be seen in a darkened room. The glow only lasts about 30 seconds but can be documented photographically. The glow is stronger in areas receiving more spray; the intensity of the glow does not indicate the amount of blood or other activator present.
- ^ Merck Index, 11th Edition, 5470.
- ^ Khan, Parvez; Idrees, Danish; MOxley, Michael A.; et al. (May 2014). "Luminol-Based Chemiluminescent Signals: Clinical and Non-clinical Application and Future Uses". Applied Biochemical Biotechnology. 173 (2): 333–355. doi:10.1007/s12010-014-0850-1. PMC 4426882. PMID 24752935.
Previous Page Next Page



